A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material required to accomplish the following transformation?
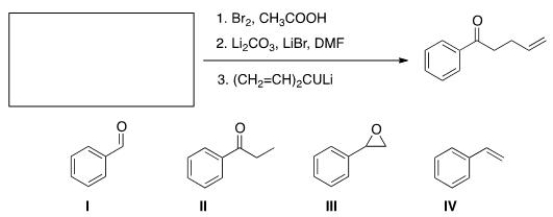
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the product(s) of the following reaction?
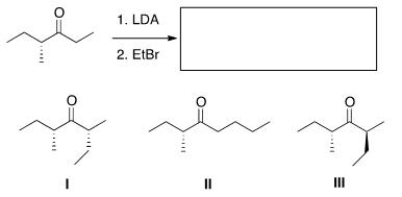
A) Only I
B) Only II
C) Only III
D) Only I and III
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an enol form of the following compound?
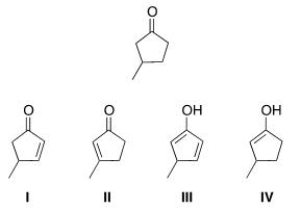
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the intermediate for halogenation of ketones under acidic conditions?
A) An enolate
B) An enol
C) A tautomer
D) An epimer
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below?
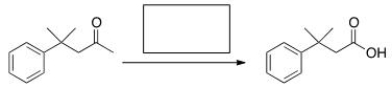
A) Br2/AcOH
B) I2/KOH
C) I2
D) KOH
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you want to form a thermodynamic enolate, you want to:
A) Keep the reaction as cold as possible.
B) Use an aprotic solvent such as THF.
C) Use a protic solvent such as ethanol.
D) Use a carboxylic acid.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would undergo racemization in the presence of a base?
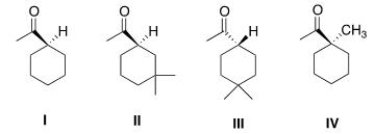
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the structure of X, product of the following reaction?
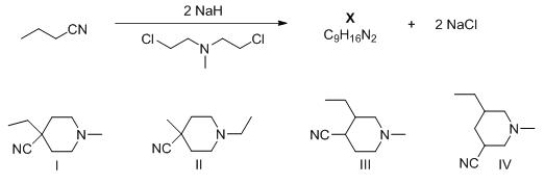
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the more stable form of acetophenone?
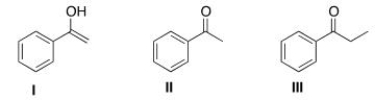
A) Only I
B) Only II
C) Only III
D) I and II are equally stable
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you want to form a kinetic enolate, you want to:
A) Use a strong, non-nucleophilic base such as LDA.
B) Use a protic solvent.
C) Use a low temperature.
D) Both use a strong, non-nucleophilic base such as LDA and use a low temperature.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction?
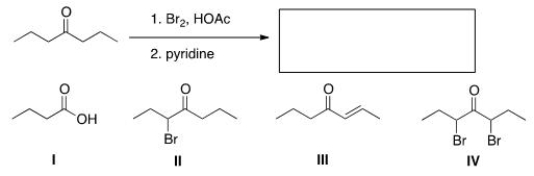
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
It has been found that b-dicarbonyl compounds have a greater concentration of the enol form over the keto form.This can be explained by:
A) The C=C of the enol is conjugated with the carbonyl group.
B) The -OH of the enol can hydrogen bond to the oxygen of the nearby carbonyl group.
C) Both A and B above are true.
D) None of the choices are true.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material for the following reaction?
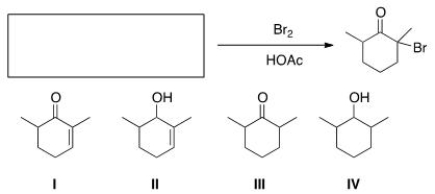
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most acidic proton in the following compound?
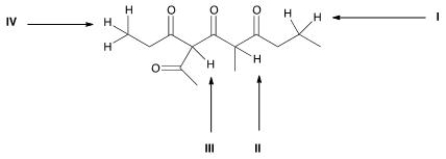
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
For most compounds with a single keto group in the molecule, equilibrium favors the keto form over the enol form of the compound.This is due largely to what?
A) The C=O bond is much stronger than the C=C bond.
B) The C=C bond is much stronger than the C=O bond.
C) The keto form can undergo intramolecular hydrogen bonding.
D) The enol form can undergo intramolecular hydrogen bonding.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following bases will completely convert 1,4-cyclohexandione into an enolate?
A) Sodium hydroxide
B) Sodium methoxide
C) Sodium tert-butoxide
D) Sodium hydride
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most acidic proton in the following compound?
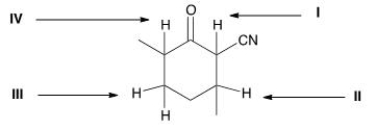
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A simple chemical test to distinguish between acetone and 3-pentanone would be the reaction of the compounds with
A) bromine with acetic acid.
B) bromine and aqueous hydroxide ion.
C) THF, LDA at -78 °C followed by reaction with bromine.
D) base and methyl bromide.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent for the following reaction?
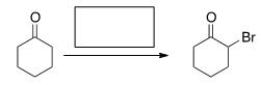
A) Br2/HOAc
B) Br2/KOH
C) Cl2/FeCl3
D) Br2/FeBr3
F) B) and D)
Correct Answer

verified
A
Correct Answer
verified
Showing 1 - 20 of 40
Related Exams