A) n
B) l
C) ml
D) ms
E) None of the quantum numbers can have any value that is not an integer.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the group of the periodic table in which an element with the following electron configuration belongs: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4
A) VA
B) IIIA
C) IVA
D) VIA
E) none of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict which atom is most likely to form a negative ion.
A) Mg
B) Si
C) S
D) Te
E) Ca
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the period and group in the periodic chart that contains the element with the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4
A) period 4, group VIA
B) period 6, group IVA
C) period 3, group IV B
D) period 4, group IVA
E) none of these
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following atoms in order of increasing first ionization energies.
A) Be < Mg < Ca < Sr
B) Mg < Ca < Sr < Be
C) Sr < Ca < Mg < Be
D) Ca < Sr < Mg < Be
E) none of these are in the correct order
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the core charge for the silicon atom, Si?
A) +1
B) +2
C) +4
D) +6
E) +8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following atoms in order of decreasing energy needed to remove the most tightly held electron (the electron closest to the nucleus) . Na, Mg, Al, P
A) Na > Mg > Al > P
B) Na > Mg > P > Al
C) Al > P > Na > Mg
D) Al > P > Mg > Na
E) P > Al > Mg > Na
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of unpaired electrons that can be accommodated in a 5d subshell?
A) 3
B) 5
C) 6
D) 7
E) 10
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons that can be accommodated in the subshell for which n = 3 and l = 2?
A) 2
B) 6
C) 10
D) 14
E) 18
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A recent article in Scientific American notes that life would be impossible on any planet close enough to the Sun to boil water, or far enough from the Sun that water froze. It also argues that the Earth is in one of the few parts of the galaxy where life could exist. Too close to the core, and either collisions with other objects would destroy the planet or cosmic radiation from neighboring stars would destroy life. Too far from the sun and there wouldn't be enough of the elements needed to form a planet. Let's assume that radiation becomes particularly dangerous to life when it carries enough energy to ionize a water molecule when it is absorbed. ?
 Use Avogadro's number and Planck's constant to calculate the frequency of this radiation to one significant figure. (Hint: Pay close attention to units!)
Use Avogadro's number and Planck's constant to calculate the frequency of this radiation to one significant figure. (Hint: Pay close attention to units!)
A) 3 x 109 s-1
B) 3 x 1012 s-1
C) 3 x 1015 s-1
D) 3 x 1018 s-1
E) 3 x 1021 s-1
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Theoreticians predict that the element with atomic number 120 will be more stable than the elements recently discovered with atomic numbers between 103 and 109. Based on the order of filling of atomic orbitals, the chemistry of this element should most closely resemble the chemistry of which of the following?
A) Ra, Group IIA
B) Pb, Group IVA
C) Po, Group VIA
D) Rn, Group VIIIA
E) one of the transition metals between La and Hg
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which transition shown below will require the absorption of the shortest wavelength photon?
A) n = 1 to n = 3
B) n = 2 to n = 3
C) n = 1 to n = 5
D) n = 8 to n = 1
E) All of the above will require the same wavelength photon.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element is represented by the following PES spectrum? 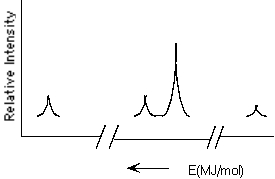
A) N
B) F
C) Na
D) Al
E) O
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following elements in order of increasing first ionization energy: S, Ar, Ca
A) S < Ar < Ca
B) Ar < S < Ca
C) Ca < Ar < S
D) S < Ca < Ar
E) Ca < S < Ar
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Water ionizes according to the following reaction which requires approximately 1200 kJ/mol of energy H2O(l) + h v H2O+ + e- In what portion of the electromagnetic spectrum are you likely to find the radiation that carries just enough energy to be dangerous to life because it can ionize the water that is so important to living organisms?
A) Radio/TV waves, 10 - 0.1 m
B) Microwaves, 0.01 - 10-4 m
C) Ultraviolet, 10-7 - 10-9 m
D) X-rays, 10-10 - 10-12 m
E) -rays, 10-12 - 10-14 m
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following describes the electron configuration for the Sn2+ ion?
A) [Kr] 4d10
B) [Kr] 5s2
C) [Kr] 5s2 5p2
D) [Kr] 5s2 4d10
E) [Kr] 5s2 4d10 5p2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In 1814 Fraunhofer observed a series of dark lines in the sun's spectrum, which he labeled A through H. About 50 years later, Gustav Kirchhoff noticed that the wavelength of light given off when sodium salts are added to a flame is the same as the wavelength of the D line in Fraunhofer's spectrum. He concluded that certain substances give off light when heated that has the same wavelength as the light absorbed under other conditions. The wavelength of the characteristic yellow-orange light emitted by sodium ions in a burner flame is 589.5923 nm. What is the energy of this light, in units of kJ/mol?
A) less than 1 kJ/mol
B) between 1 and 10 kJ/mol
C) between 10 and 100 kJ/mol
D) between 100 and 1000 kJ/mol
E) more than 1000 kJ/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
O2 molecules can dissociate to form O atoms by absorbing electromagnetic radiation. If it takes 498 kJ to dissociate one mole of O2 molecules to form two moles of O atoms, in what portion of the electromagnetic spectrum would light have sufficient energy to cause this reaction to occur?
A) radio wave
B) microwave
C) infrared
D) visible
E) ultraviolet
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What do we mean when we say that "The energy of the electron in an atom is quantized?"
A) The electron has a very small amount of energy.
B) The energy of the electron is proportional to the mass of the nucleus.
C) When an electron changes its energy, it emits a quantum of light.
D) The energy of the electron can have certain fixed energies and not others.
E) The electron must be a wave.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atom contains the largest number of unpaired electrons?
A) B
B) N
C) F
D) Ti
E) Cu
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 106
Related Exams