A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would you expect to have ionic bonds?
A) CO
B) FBr
C) NF3
D) NaCl
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of the condensed structure, CH3COCH3, to a Lewis structure? 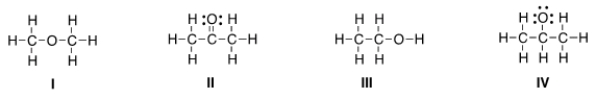
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has a net dipole moment of zero? 
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is more important in each pair of contributing resonance structures? 
A) II, IV, V
B) II, III, V
C) II, III, VI
D) I, IV, V
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules does not have a net dipole moment of zero?
A) CCl4
B) BF3
C) CO2
D) NH3
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of (CH3) 2CHOCH2CH2CH2OH to a skeletal structure? 
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C2H6O?
A) 1
B) 2
C) 3
D) 4
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in decreasing order of ionic character, putting the most ionic first.
A) I > II > III > IV
B) IV > II > I > III
C) IV > III > II > I
D) IV > II > III > I
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about valence electrons is true?
A) They are the most tightly held electrons.
B) They do not participate in chemical reactions.
C) They are the outermost electrons.
D) They reveal the period number of a second-row element.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 70 of 70
Related Exams