A) P and O
B) Al and Cl
C) Nb and Ar
D) Ag and Bi
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Characterize EACH of the following statements as being TRUE or FALSE and then indicate the collective true-false status using the choices provided. (1) Most of the mass of an atom is concentrated in its nucleus. (2) Sufficient information is present on a "standard" periodic table to determine the number of neutrons present in atoms of an element. (3) An atom whose electron configuration is 1s22s22p63s1 contains 12 electrons.
A) All three statements are true.
B) Two of the three statements are true.
C) Only one of the statements is true.
D) None of the statements is true.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isotopes of a given element
A) have the same mass number but different numbers of protons
B) have different mass numbers but identical numbers of protons
C) have the same atomic number but different chemical properties
D) have the same mass number but different chemical properties
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements concerning the nucleus of an atom is correct?
A) It accounts for almost all of the mass of an atom.
B) It is always positively charged.
C) It contains only neutrons.
D) More than one correct response.
E) No correct response.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a d subshell is correct?
A) It is found only in shells greater than 2.
B) It contains 3 orbitals.
C) It is found only in shells 2 and 3.
D) It is found in all shells greater than 1.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is a transition element?
A) (9F)
B) (50Sn)
C) (79Au)
D) (92U)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Characterize EACH of the following statements as being TRUE or FALSE and then indicate the collective true-false status using the choices provided. (1) The number of elements that are metals and nonmetals is approximately equal. (2) The number of orbitals present in an electron subshell is always six. (3) An atom as a whole is electrically neutral because all charged subatomic particles reside in the nucleus.
A) All three statements are true.
B) Two of the three statements are true.
C) Only one of the statements is true.
D) None of the statements is true.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The nuclear charge for an atom of 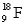 is
is
A) zero
B) + 9
C) + 18
D) + 28
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the appropriate electron configuration for an element that has two unpaired electrons.
A) 1s22s22p63s23p6
B) 1s22s22p63s23p4
C) 1s22s22p63s1
D) 1s22s22p5
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Characterize EACH of the following statements as being TRUE or FALSE and then indicate the collective true-false status using the choices provided. (1) The shape of an s orbital is always spherical. (2) All naturally occurring atoms of an element must be identical to each other in terms of subatomic particle makeup. (3) The alkaline earth metals are the elements in group IA of the periodic table.
A) All three statements are true.
B) Two of the three statements are true.
C) Only one of the statements is true.
D) None of the statements is true.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following electron configurations corresponds to an element in the same group of the periodic table as the element whose electron configuration is 1s22s22p5?
A) 1s22s22p4
B) 1s22s22p6
C) 1s22s22p63s23p64s23d5
D) more than one correct response
E) no correct response
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
After the 4s subshell of an atom is filled with electrons, the next electron added will enter the
A) 3d subshell
B) 4p subshell
C) 4d subshell
D) 5s subshell
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Characterize EACH of the following statements as being TRUE or FALSE and then indicate the collective true-false status using the choices provided. (1) The maximum number of electrons that can occupy an electron shell is the same as the shell number. (2) A nucleus is the very small dense uncharged center of an atom. (3) The number of inner transition elements is greater than the number of transition elements.
A) All three statements are true.
B) Two of the three statements are true.
C) Only one of the statements is true.
D) None of the statements is true.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the appropriate pair of atoms that contain the same number of protons.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chlorine, which exists in nature in two isotopic forms, has an atomic mass of 35.5 amu. This means that:
A) All chlorine atoms have masses of 35.5 amu.
B) More than half of all chlorine atoms have masses of 35.5.
C) 35.5 amu is the upper limit for the mass of a chlorine atom.
D) The mass of one isotope must be lighter than 35.5 amu while the mass of the other isotope is heavier than 35.5 amu.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following element classification pairings is incorrect?
A) O representative element
B) Xe noble gas element
C) Cr transition element
D) Os inner transition element
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is an inner-transition element?
A) (9F)
B) (50Sn)
C) (79Au)
D) (92U)
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is correct for 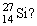
A) atomic number = 27 and mass number = 14
B) contains 14 protons and 27 neutrons
C) contains an equal number of protons and electrons
D) more than one correct response
E) no correct response
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the correct atom that has a mass number of 17.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Period number and group number in the periodic table are numerically equal for which of the following elements?
A) (13Al)
B) (33As)
C) (51Sb)
D) more than one correct response
E) no correct response
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 70
Related Exams